With increasing consumption, food products and trading are also on the rise. The quality and safety of food products are increasingly considered as factors in consumer protection. Many countries have regulations for the import and export of food products. Indonesia is one of the countries in the world with the largest population and is of course is a large market for the food industry.
Some approvals required before the food product can be marketed in Indonesia are Halal, SNI, product registration at National Food and Drug Agency and product registration at Ministry of Trade.
Halal certification is important in Indonesia as the majority of the population are Muslim. Regarding SNI approval for food products, at the moment there are 11 products that have been mandatory such as packed drinking water (SNI 01-3553-2006), natural mineral water (SNI 01-6242-2000), raw sugar (SNI 01.3140-1:2001), refined sugar (SNI 01.3140-2:2006), white sugar (SNI 01.3140-3:2010), iodized salt (SNI 01-3556-2000), wheat flour (SNI 3751:2009), cocoa powder (SNI 3747:2009), biscuit (SNI 2973:2011), instant coffee (SNI 2983:2014), and palm cooking oil (SNI 7709:2012).
TÜV Rheinland Indonesia has been accredited for nine of these products which are packed drinking water (SNI 01-3553-2006), natural mineral water (SNI 01-6242-2000), raw sugar (SNI 01.3140-1:2001), refined sugar (SNI 01.3140-2:2006), wheat flour (SNI 3751:2009), cocoa powder (SNI 3747:2009), biscuit (SNI 2973:2011), instant coffee (SNI 2983:2014), and palm cooking oil (SNI 7709:2012).
Regarding the SNI certification process, all food products need to comply by the type 5 product certification scheme. This scheme requires a factory audit, sample drawing and product testing. During the audit, it will assess the management system, production process and implementation of Good Manufacturing Practices (GMP). For product testing, sampling will need to be taken at the production line or factory warehouse.
The following table shows the family for each product category and number of samples to be taken.
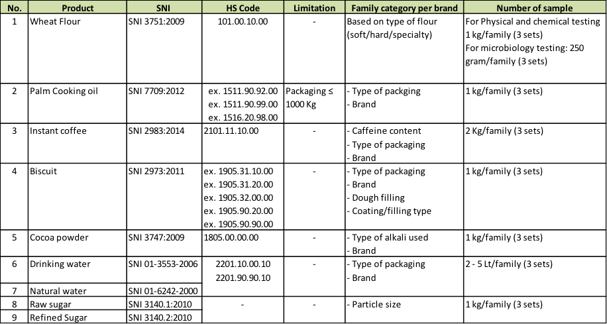
For food products, there are three main parameters needed for testing which are physical such as taste, odour, appearance, etc; chemical such as free fatty acid, heavy metals, etc; and microbiological such as yeast and mold, TPC, E.coli, etc. All products will be tested based on SNI and at appointed testing laboratories in Indonesia. For product testing it will take between 10 – 15 working days.
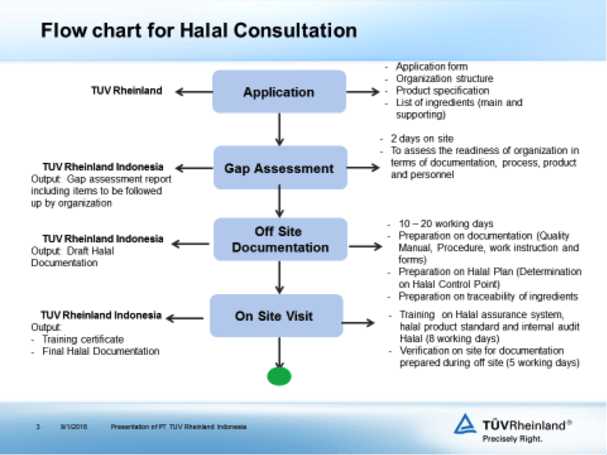
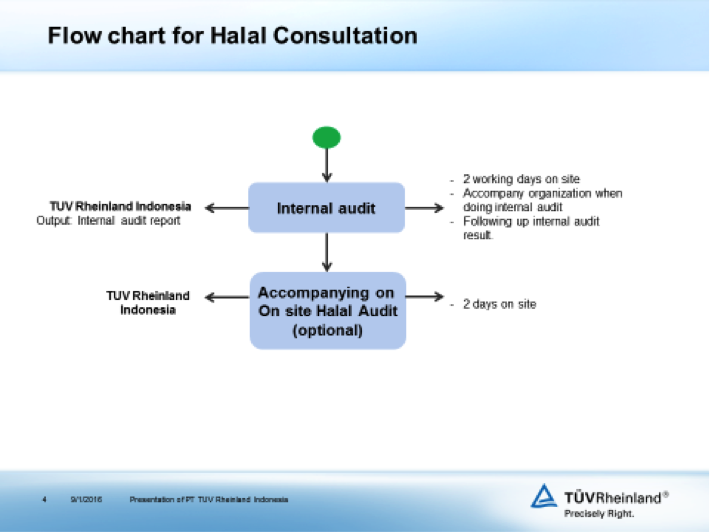
Regarding Halal certification, TÜV Rheinland Indonesia can assist the food industry to get certification from LPPOM MUI. The following is the flowchart for Halal consultation.
After getting SNI certification especially for mandatory products and Halal certification, the food manufacturer needs to register their product to the National Food and Drug Agency to get an ML number. Several documents need to be submitted for ML registration as follows:
- Original agency letter/letter of appointment
- Letter that states/explains the relationship between the company in Indonesia and the principal in overseas. The letter has an issue and expiry date.
- Original health certificate/free sale
- Health certificate for the product issued by the authority in the country of origin.
- Product specification that includes 100% fixed ingredients
- Specification of raw materials
- Information about shelf life of product
- Production flow chart
- Explanation on production code
- Specification of type of packaging
- Certificate of Analysis of the product from accredited testing laboratory in Indonesia (ISO/IEC 17025) or overseas that has MLA APLAC/ILAC
- Soft label/label design of the marketed product in Indonesia
- Sample minimal 3 pcs (for the sample of testing in Indonesia and verification of product photo)
The registration process can be done online via: www.e-reg.pom.go.id. Before accessing this website, the company needs to have an ID. To get the ID, they must register by visiting BPOM and submitting the following documents:
- Importer identification number (API) for importer
- IT for importer
- PSB factory/warehouse that is issued by audit team from National Food and Drug Agency
In order to get PSB (Inspection on facility), the company musts submit the following documents:
- Application letter
- SIUP (Surat Ijin Usaha Perusahaan) – shall be for food and beverage
- NPWP (Nomor Pokok Wajib Pajak) – Tax number
- TDP (Tanda Daftar Perusahaan)
- Domicile letter
- Notaries Act
- Location map
- Facility map
- Certificate of Analysis
- SOP for hygiene and sanitation
- SOP for receiving product
- SOP for pest control
- Organizational structure
- Copy of Importer Identification Number (API)
- Original free sale/health certificate
- LOA (letter of acceptance from manufacturer as distributor in Indonesia)
- Photos of Fire extinguisher (mandatory)
- Photos of Pallet/rack for storage (mandatory)
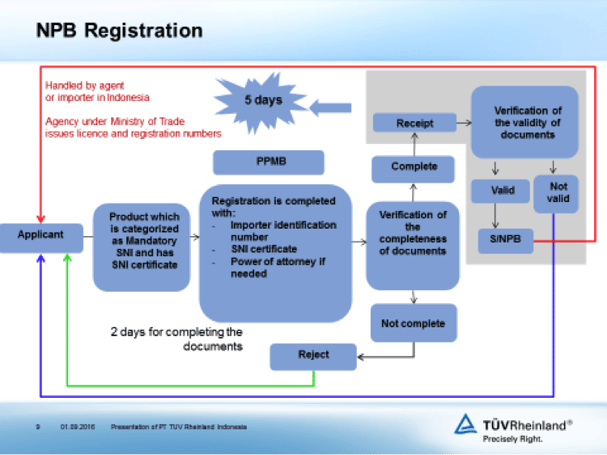
With regards to market surveillance for products that have been mandatory of SNI, the Ministry of Trade requires the importer to register their product in term of NPB number. Below is the NPB registration flowchart.
Product labeling is also one of issues for imported products that are marketed in Indonesia. For food products, labels must follow the requirements on Government Regulation No. 69 Year 1999. The following is the information to be put on the label:
- Name of product
- List of ingredients
- Nett weight
- Name and address of importer
- Expiry date
- Registration number
- Production code
- Nutritional information
- Information of food irradiation and GMO (if any)
- Information on natural raw material (if any)
- Information on intended use for immunodeficiency people (if any)
- Information on food additives
The label must be written in Bahasa Indonesia and can include Arabic or Latin numbers.
For more information on our Food Testing solutions, please visit our website or contact us to speak to one of our experts.



